
On a day dominated by global volatility and energy-driven headlines, a small Australian biotech quietly rewrote the script.
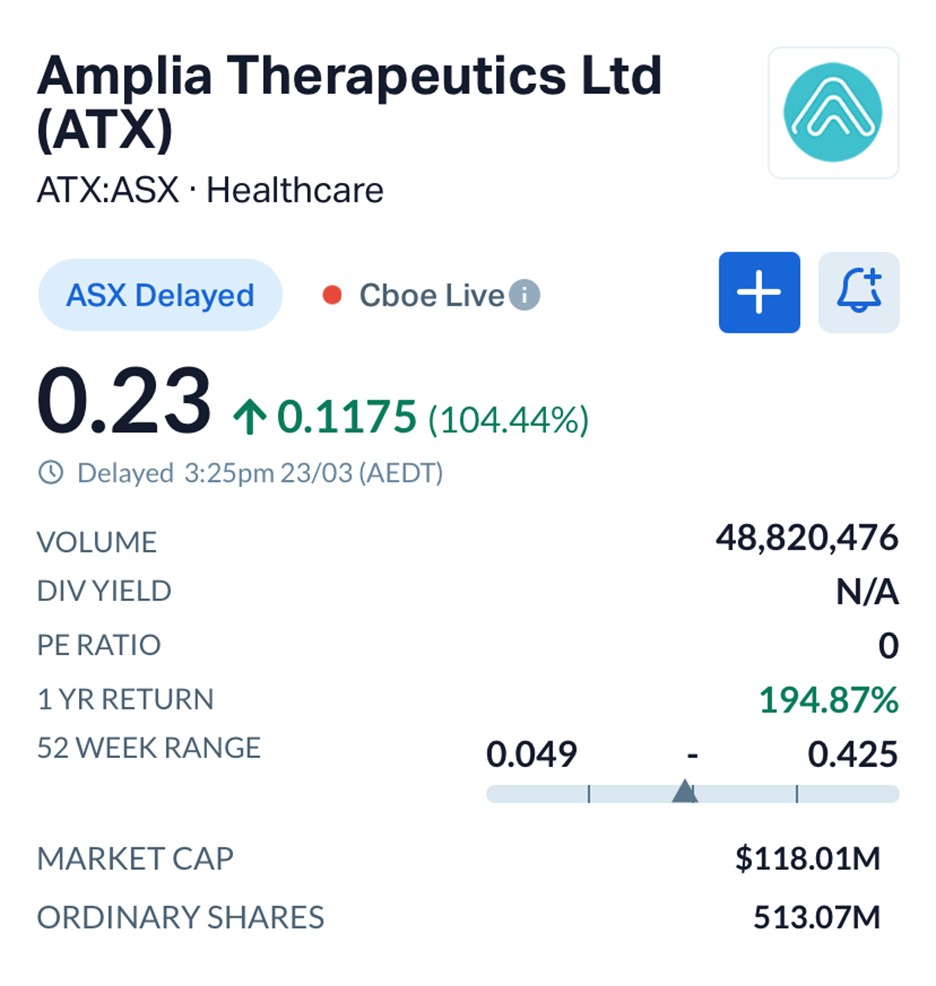
Amplia Therapeutics (ASX: ATX) surged 104.44% to $0.23, with more than 46 million shares traded, after releasing updated clinical data from its ACCENT trial in advanced pancreatic cancer.
Source: MarketIndex
This was not a typical biotech rally. It was a re-rating event.
When a stock doubles in a single session on clinical results, it usually signals a shift in how the market views the company. Amplia is no longer being treated as an early-stage hopeful. It is now being priced as a credible mid-stage contender with global relevance.
At the heart of the surge lies one number: 7.8%.
That is the Complete Response rate recorded in Amplia’s trial. In oncology terms, a Complete Response means the total disappearance of measurable tumours and metastases for at least two months.
To put that into perspective, historical trials for standard chemotherapy treatments such as those in the MPACT and NAPOLI 3 studies have shown Complete Response rates of just 0.2% to 0.3%.
The gap is striking.
It suggests that Amplia’s lead drug, narmafotinib, is not simply improving outcomes at the margin. It may be fundamentally altering how patients respond to treatment.
The trial also reported a median Overall Survival of 11.1 months, compared to the typical 8.5 to 9.2 months seen with chemotherapy alone.
In isolation, a two-month improvement may sound modest. But in pancreatic cancer, one of the most aggressive and difficult-to-treat cancers, this is a meaningful leap.
Historically, incremental gains in survival have been the benchmark for regulatory approval. What makes Amplia’s data particularly compelling is that this improvement was achieved without increasing toxicity.
Patients are not just living longer. They are doing so without additional side effects.
Beyond Complete Responses, the Objective Response Rate was updated to 35.9%, meaning more than one in three patients experienced a measurable reduction in tumour size or complete disappearance.
This places Amplia’s results firmly within, and in some cases above, the upper range of global benchmarks for mid-stage trials in pancreatic cancer.
Importantly, these findings were not solely based on internal assessments. The data underwent independent central review using RECIST 1.1 criteria, the gold standard for evaluating tumour response.
This step removes potential bias and is often a prerequisite for advancing into late-stage trials or attracting pharmaceutical partnerships.
Amplia CEO Dr Chris Burns emphasised the broader significance of the findings.
“These latest data from the ACCENT trial clearly demonstrate the significant clinical benefit of narmafotinib. The unprecedented 7.8% rate of CR’s in the first line setting provides new hope for patients with this very aggressive cancer and provides further strong support for the benefit that narmafotinib can bring when combined with other treatment modalities. We look forward to presenting a detailed analysis of the ACCENT trial at the forthcoming AACR conference.”
Pancreatic cancer has long been one of the most challenging areas in oncology, with limited progress over the past two decades.
According to research published in the New England Journal of Medicine and The Lancet, survival outcomes have remained stubbornly low despite multiple treatment advances.
Against that backdrop, Amplia’s data stands out.
It matches the survival performance of more complex, recently approved combination therapies such as NALIRIFOX, while maintaining a simpler and potentially more tolerable treatment profile.
That combination of efficacy and safety is what global pharmaceutical companies look for when evaluating acquisition or licensing opportunities.
Amplia has also been selected to present its data at the American Association of Cancer Research conference in April 2026 in San Diego.
This is not a routine presentation.
The AACR is one of the most influential gatherings in the oncology world. Selection signals scientific credibility and places the company directly in front of major pharmaceutical players.
For a company with a market capitalisation of approximately $118 million, the exposure could be transformative.
The scale of the share price move reflects more than just strong data.
It reflects a shift in perception.
Biotech investing often follows a binary pattern. Companies are either viewed as speculative or validated. Amplia’s latest results appear to have pushed it firmly into the latter category.
The surge in trading volume suggests that institutional capital is beginning to take notice, not just retail participants reacting to headlines.
The immediate focus will be on deeper analysis of the ACCENT trial data and its presentation at AACR.
Beyond that, the path leads toward larger, registrational trials that could support regulatory approval.
At the same time, the company’s drug, narmafotinib, may attract interest from larger pharmaceutical groups seeking to expand their oncology pipelines.
Amplia Therapeutics ASX Announcement, March 23, 2026; New England Journal of Medicine (MPACT Study); The Lancet (NAPOLI 3 Trial)
Disclaimer - Skrill Network is designed solely for educational and informational use. The content on this website should not be considered as investment advice or a directive. Before making any investment choices, it is crucial to carry out your own research, taking into account your individual investment objectives and personal situation. If you're considering investment decisions influenced by the information on this website, you should either seek independent financial counsel from a qualified expert or independently verify and research the information.
Tags:
RECENT POSTS
TAGS
Subscribe to the Skrill Network Newsletter today and stay informed
Recommended Articles