Lumos Diagnostics has secured a key US regulatory approval and fresh capital, marking what many would describe as a defining pivot from development to commercial scale.
For healthcare companies, regulatory approval in the United States is often the single biggest hurdle. Lumos has now cleared it.
The company confirmed that its flagship FebriDx test has received a CLIA waiver from the US Food and Drug Administration. In simple terms, this means the test can now be used outside traditional laboratories.
Instead of waiting days for results, doctors can diagnose patients in minutes during a consultation.
That shift is significant.
It expands Lumos’ reach from a limited lab-based market to more than 300,000 decentralised healthcare settings, including GP clinics, urgent care centres and pharmacies across the US.
The company estimates this opens access to around 80 million patients annually, effectively increasing its addressable market by 15 times.
CEO Doug Ward framed the milestone clearly, saying:
“The granting of a CLIA waiver for FebriDx by the US FDA marks a transformative moment for the management of acute respiratory infections in the US healthcare system. This approval allows Lumos to access a vastly larger market, allowing healthcare professionals in outpatient clinics to deliver rapid and accurate results at the time of consultation to deliver more effective patient outcomes.”
The appeal of FebriDx lies in its simplicity.
The test helps distinguish between bacterial and viral infections in about 10 minutes, a distinction that is critical in deciding whether antibiotics are needed.
Traditionally, this process can take days, often leading to precautionary antibiotic use.
That inefficiency has contributed to one of healthcare’s biggest long-term challenges, antibiotic resistance.
By moving diagnostics to the point of care, Lumos is positioning itself within a broader global push toward faster, cheaper and more precise treatment pathways.
In effect, the company is tapping into what industry analysts often call the “point-of-care revolution,” where testing shifts from centralised labs to frontline healthcare settings.
Alongside the regulatory milestone, Lumos has strengthened its balance sheet with a A$20 million institutional placement.
Demand for the raise exceeded expectations, signalling strong backing from both existing and new investors.
An additional A$3.1 million is expected from option exercises by major shareholders, bringing total committed funding to A$23.1 million.
The placement was priced at A$0.225 per share, representing a 15 percent discount to the last closing price.
Participants also received free-attaching options, a structure commonly used to incentivise longer-term support.
The company is also launching a Share Purchase Plan to raise up to A$2 million, giving retail shareholders an opportunity to participate at the same price.
Doug Ward highlighted the strategic intent behind the capital raise:
“This additional capital strengthens Lumos’ balance sheet to scale up our manufacturing, sales and marketing capacity to commence the broader commercialisation of FebriDx in the United States. We are grateful for the commitment of these new investors and our existing shareholders, whose support is pivotal in enabling us to execute on our strategy and maintain momentum at this important inflection point.”
The funding is not about survival. It is about execution.
Lumos plans to deploy the capital across several key areas:
This signals a transition from a research-focused company to a commercially driven healthcare business.
The US remains the largest diagnostics market globally, and Lumos is now positioned to compete in a segment estimated to exceed US$1 billion.
The timing of this announcement is notable.
Healthcare systems globally are under pressure to reduce costs while improving outcomes.
Rapid diagnostic tools like FebriDx address both challenges.
At the same time, post-pandemic healthcare models are increasingly shifting toward decentralised care, where patients are treated faster and closer to home.
This structural trend aligns directly with Lumos’ strategy.
Shares in Lumos were trading at $0.260 as of early afternoon, holding steady despite broader market volatility.
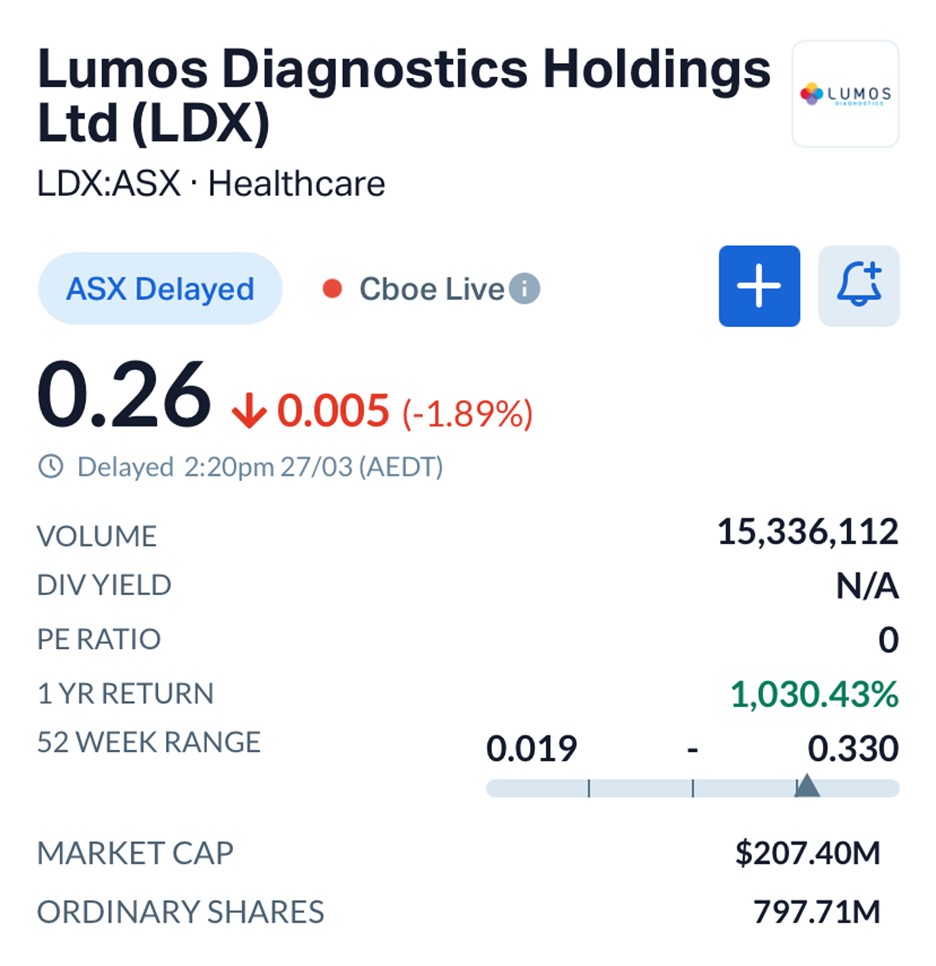
Source: MarketIndex
The stock has delivered a remarkable one-year return of over 1,000 percent, reflecting growing confidence in its commercial pathway.
Still, the real test lies ahead.
Execution risk remains, particularly in scaling distribution and achieving widespread adoption in the US healthcare system.
Lumos’ update is more than just a regulatory win.
It represents a shift in identity.
From a company developing diagnostic tools, it is now stepping into the role of a commercial healthcare player targeting one of the world’s most competitive markets.
In a week where global markets are grappling with uncertainty, Lumos offers a different narrative.
One built on regulatory validation, capital backing, and a clear pathway to revenue.
Whether that translates into sustained growth will depend on how effectively it can turn this momentum into market share.
Disclaimer - Skrill Network is designed solely for educational and informational use. The content on this website should not be considered as investment advice or a directive. Before making any investment choices, it is crucial to carry out your own research, taking into account your individual investment objectives and personal situation. If you're considering investment decisions influenced by the information on this website, you should either seek independent financial counsel from a qualified expert or independently verify and research the information.
Tags:
RECENT POSTS
TAGS
Subscribe to the Skrill Network Newsletter today and stay informed
Recommended Articles