
In an industry known for burning cash and relying on investors to survive, Neuren Pharmaceuticals Ltd is doing something unusual. It is generating enough revenue from its existing drug to fund the next generation of treatments on its own.
The company reported $65 million in royalty income from its Rett syndrome treatment DAYBUE, marking a 15 percent increase and reinforcing its position as one of the rare profitable biotechnology firms on the ASX.
The steady income stream has helped Neuren build a cash balance of $296 million, giving it financial independence at a time when many biotech peers are struggling to secure funding.
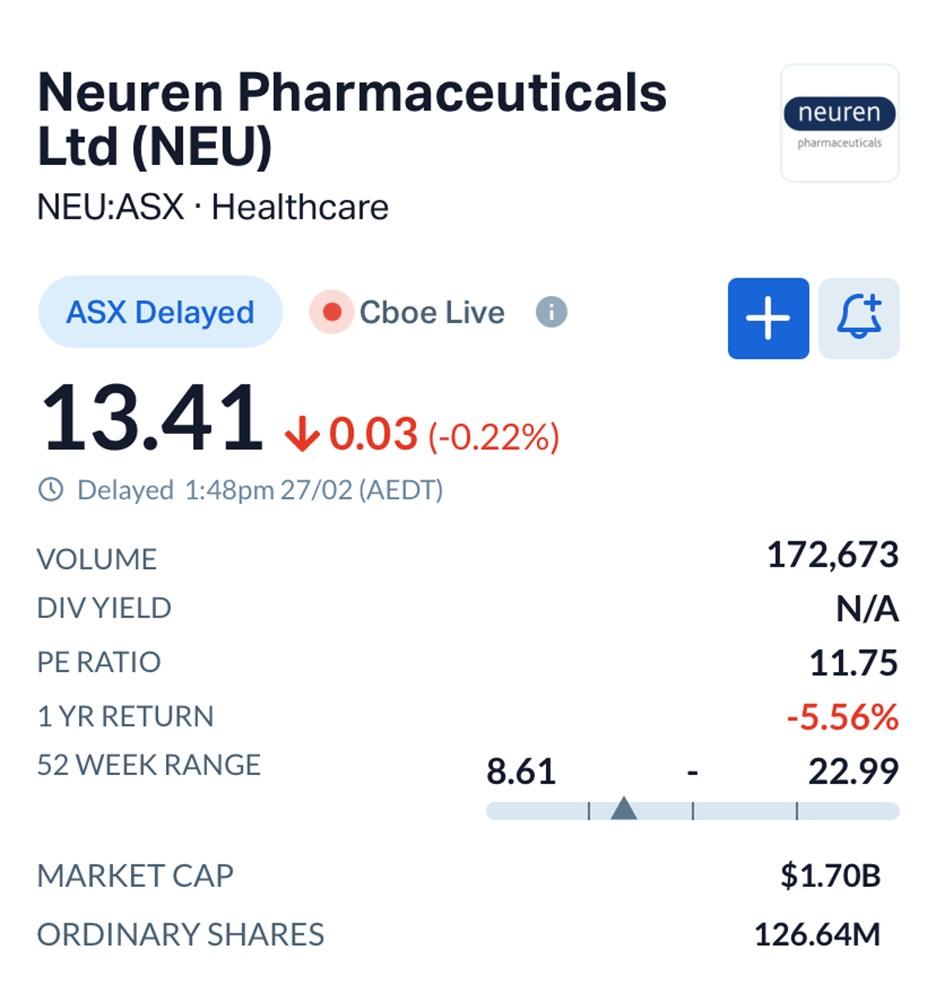
By early afternoon trading on Friday, Neuren shares were holding steady at $13.41, giving the company a market value of about $1.7 billion, according to ASX data.
Source: MarketIndex
Biotechnology companies typically operate at a loss for years while funding expensive clinical trials. Neuren’s trajectory has been different.
Its breakthrough came through DAYBUE, a treatment developed for Rett syndrome, a rare neurological disorder that affects brain development and primarily impacts young girls.
DAYBUE generates royalty income for Neuren through its commercial partner, creating a recurring revenue stream. That income is now funding the company’s research pipeline, eliminating the need for frequent capital raisings that dilute shareholders.
Chief executive officer Jon Pilcher highlighted the significance of this shift, noting that the company’s clinical programs are fully funded internally.
“All of our current clinical trials are self funded by DAYBUE revenue,” Pilcher said, underscoring the financial independence the company has achieved.
This makes Neuren what analysts often call a “self sustaining biotech”, a rare model in a sector where companies usually depend heavily on external financing.
While DAYBUE provides stable income, the company’s future growth may depend on its next drug candidate, NNZ 2591.
Neuren confirmed it has begun dosing patients in its Phase 3 “Koala” trial, targeting Phelan McDermid syndrome, another rare neurological disorder.
Phase 3 trials are the final and most critical stage before a drug can be approved by regulators. Success at this stage can transform a company’s long term value.
The drug has also received Rare Pediatric Disease designation from the US Food and Drug Administration. If approved, Neuren could receive a Priority Review Voucher, which the company says has historically sold for more than US$200 million, equivalent to over A$300 million.
These vouchers allow pharmaceutical companies to accelerate the approval of other drugs and are highly sought after in the industry.
Neuren is also working to extend the reach of DAYBUE itself.
The US Food and Drug Administration has approved a new powder formulation called DAYBUE STIX. This version is designed to help patients who struggle with the original liquid format.
The new formulation could expand access significantly. According to company data, a large portion of eligible patients have not yet started treatment, suggesting room for further growth.
The company is also pursuing approval in Europe. While regulators there have raised questions, Neuren has requested a re-examination of its application.
Importantly, the company’s royalty guidance of $70 million to $77 million for 2026 does not include European sales, meaning any approval would add additional upside.
In another signal of confidence, Neuren announced it will begin a share buyback program starting March 2, 2026.
Share buybacks reduce the number of shares in circulation, increasing each remaining investor’s ownership stake and often supporting share prices.
For many companies, buybacks are a sign that management believes their stock is undervalued and that future earnings are secure.
This move reinforces Neuren’s transformation from a speculative biotech into a profitable pharmaceutical business with stable cash flows.
Neuren’s model stands out in the biotechnology sector.
According to industry research, most biotech companies operate without revenue for years while developing treatments. Only a small percentage successfully reach profitability through commercialized drugs.
Neuren’s ability to generate revenue and reinvest it into research places it among a select group of self funding biotechnology firms.
This structure reduces financial risk and allows the company to focus on developing new therapies without relying on external capital.
Neuren’s success with DAYBUE has reshaped its financial position and future trajectory.
Royalty income is funding its research pipeline, its cash reserves provide stability, and its Phase 3 trial represents a potential next wave of growth.
The company’s progress highlights a rare transition in biotechnology. It is no longer a company seeking funding to survive. It is a profitable drug developer using its own earnings to build its future.
Source: Neuren Pharmaceuticals financial update, FDA approval data, ASX market data
Disclaimer - Skrill Network is designed solely for educational and informational use. The content on this website should not be considered as investment advice or a directive. Before making any investment choices, it is crucial to carry out your own research, taking into account your individual investment objectives and personal situation. If you're considering investment decisions influenced by the information on this website, you should either seek independent financial counsel from a qualified expert or independently verify and research the information.
Tags:
RECENT POSTS
TAGS
Subscribe to the Skrill Network Newsletter today and stay informed
Recommended Articles