In a hospital room in Hong Kong, far from trading floors and market noise, a small but significant step unfolded.
Racura Oncology has successfully dosed its third patient in a Phase 1 cancer trial, marking the completion of its initial safety cohort for RC220, a drug designed to do something rare in oncology. Kill cancer while protecting the heart.
For a company operating in the high-risk world of biotech, this moment represents more than progress. It signals a transition from theory to human validation.
Early-stage clinical trials often pass quietly, but within the industry, the first successful human dosing phase is closely watched.
Racura confirmed that all three patients in the initial cohort were treated at 40mg/m² with zero dose-limiting toxicities and no phlebitis, a common and painful side effect associated with similar drugs.
That detail matters.
Historically, drugs in this class have struggled with vein irritation and safety concerns. By avoiding these issues, Racura has cleared a key technical hurdle that has limited earlier therapies.
Chief Executive Officer Dr Daniel Tillett acknowledged the significance:
“The safe dosing of the third patient in our RC220 solid tumour trial in Hong Kong and recruitment of the first dose escalation cohort is an important milestone for Racura Oncology. We are grateful to all the patients, investigators, and clinical teams who have made this trial possible and we look forward to treating patients on the updated protocol.”
With the first cohort now complete, the trial moves into its next phase.
A Safety Review Committee will assess the data before allowing escalation to the next dose level of 80mg/m², effectively doubling the current dosage.
In clinical terms, this is where the real test begins.
Phase 1 trials are designed to answer a simple but critical question. How much of the drug can patients safely tolerate? The higher the safe dose, the greater the potential for therapeutic impact.
The planned jump suggests early confidence in RC220’s tolerability profile.
At the heart of Racura’s approach is what it calls the Cardioprotection and Anticancer Synergy (CPACS) strategy.
Most chemotherapy drugs, including widely used agents like doxorubicin, are effective but come at a cost. They can cause lasting heart damage, limiting how aggressively doctors can treat cancer.
RC220 is designed to change that equation.
By combining anti-cancer activity with heart protection, the drug aims to expand treatment options for patients who might otherwise face difficult trade-offs.
If successful, it could address a longstanding gap in oncology care.
The study is being conducted across multiple sites in Australia, Hong Kong, and South Korea, reflecting a growing trend toward globally integrated clinical trials.
It also uses a Bayesian design, a more flexible approach that allows researchers to adapt the trial as data emerges. This can potentially speed up development timelines and improve decision-making.
For a small-cap biotech, this level of sophistication signals operational maturity.
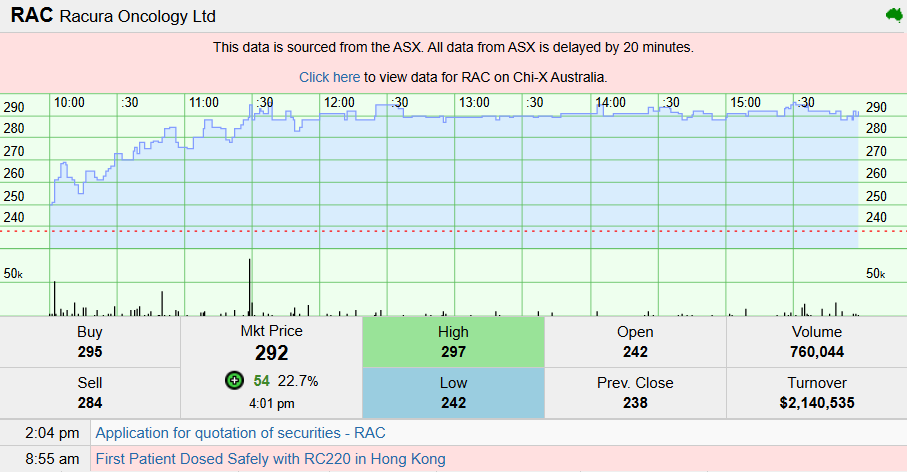
The market has responded swiftly.
Racura shares rose 21% to $2.88, with the company now carrying a market capitalisation of approximately $523 million.
The stock has delivered a 144% return over the past year, reflecting growing attention on its pipeline and clinical progress.
In a sector often driven by binary outcomes, even early-stage milestones can reshape sentiment.
Source: StocknessMonster
The broader oncology landscape is evolving.
According to global health data, cancer remains one of the leading causes of death worldwide, with millions of new cases diagnosed each year. At the same time, the focus is shifting toward targeted therapies and improved quality of life during treatment.
Drugs that can reduce side effects while maintaining effectiveness are increasingly seen as the next frontier.
Racura’s RC220 sits within this trend, aiming not just to treat cancer, but to make treatment safer.
The immediate focus is on the Safety Review Committee’s decision.
If approved, recruitment for the next cohort will begin across multiple international sites, with higher doses and broader patient data.
Further down the line, the trial will expand into additional patients to assess early signs of efficacy alongside safety.
For now, the company has achieved what many early-stage biotech firms strive for but do not always reach. A clean start in humans.
While markets reacted strongly to the update, the deeper story lies elsewhere.
Behind every data point is a patient, a treatment attempt, and a step toward something that could reshape how cancer is managed.
For Racura, this milestone is not the finish line. It is the beginning of a much longer journey.
Source:Racura Oncology ASX Announcement, Market Data, March 19, 2026
Disclaimer - Skrill Network is designed solely for educational and informational use. The content on this website should not be considered as investment advice or a directive. Before making any investment choices, it is crucial to carry out your own research, taking into account your individual investment objectives and personal situation. If you're considering investment decisions influenced by the information on this website, you should either seek independent financial counsel from a qualified expert or independently verify and research the information.
Tags:
RECENT POSTS
TAGS
Subscribe to the Skrill Network Newsletter today and stay informed
Recommended Articles